5.2.3 Host plant volatiles attractive to bark beetles
Host volatiles are attractive to a number of forest scolytids
including species in the genera Scolytus, Dendroctonus, Hylurgops,
Trypodendron and Tomicus (Goeden and Norris, 1964; Rudinsky, 1966;
Meyer and Norris, 1967a; Moeck, 1970; Byers et al., 1985; Lanne et
al., 1987; Volz, 1988; Lindelöw et al., 1992; Hobson et al., 1993).
Species of bark beetle that regularly attack and kill living trees
(termed "aggressive") have been shown invariably to possess an
aggregation pheromone, usually of two or more components, but are
weakly, if at all, attracted by host volatiles alone (Vité and
Pitman, 1969; Byers, 1989a). However, so-called "secondary" bark
beetle species (those that arrive later after the tree has already
been killed by the aggressive bark beetles or that feed as
saprophytes in decaying trees) may not use an aggregation
pheromone, but generally are strongly attracted to either host
monoterpenes, ethanol or a combination (Kohnle, 1985; Klimetzek et
al., 1986; Schroeder, 1988; Schroeder and Lindelöw, 1989).
Ethanol, probably released by microorganisms in decaying woody
tissue (Graham, 1968; Moeck, 1970; Cade et al., 1970) and stressed
plants (Kimmerer and Kozlowski, 1982), is attractive to a wide
variety of species of bark beetles (Moeck, 1970, 1981; Magema et
al., 1982; Montgomery and Wargo, 1983; Kohnle, 1985; Klimetzek et
al., 1986; Schroeder 1987, 1988; Schroeder and Eidmann, 1987;
Phillips et al., 1988; Volz, 1988; Chénier and Philogčne, 1989;
Schroeder and Lindelöw, 1989; Byers, 1992a). Primary alcohols other
than ethanol have not been reported as being attractive to
scolytids. However, only a few studies have tested methanol (Moeck,
1970; Montgomery and Wargo, 1983; Byers, 1992a); longer chain
alcohols up to hexanol did not attract Scolytids in Sweden when
they were known to be flying (Byers, 1992a). Electroantennogram
(EAG) responses of T. piniperda to a series of straight-chain
alcohols indicated that the antennae respond increasingly with
longer chain length up to a maximum between pentanol and heptanol
and then decrease (Lanne et al., 1987). The response spectrum could
be due in part to differences in volatility. Thus, although ethanol
plays a role in host selection (discussed subsequently), the EAG
response for ethanol is lower than for longer-chain alcohols (which
probably are not involved in behavior). Ethanol and CO2 are the
usual end products of sugar fermentation by microorganisms whereas
methanol is not, which probably explains the evolution of the use
of ethanol by forest insects. Moeck (1970) found methanol to be a
minor constituent and ethanol a major constituent of extracts from
Douglas-fir sapwood attractive to T. lineatum.
Various tree monoterpenes (e.g. alpha-pinene, myrcene,
terpinolene, B-pinene, Fig. 4) and turpentine are also attractive
to a large number of bark beetle species (Byers et al., 1985,
1992a; Phillips et al., 1988; Schroeder, 1988; Chénier and
Philogčne, 1989; Schroeder and Lindelöw, 1989; Miller and Borden,
1990; Phillips, 1990; Hobson et al., 1993).
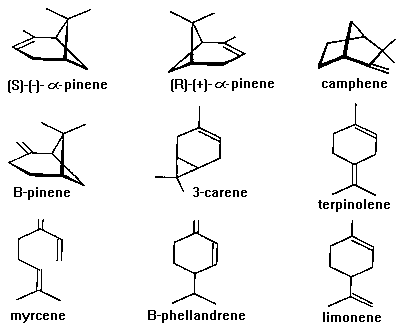
Fig. 4. Major monoterpenes of conifers. Note that the enantiomers
of alpha-pinene are identical except that they are non-superimposable
(mirror images). Camphene, B-pinene, 3-carene, B-phellandrene, and
limonene also have two enantiomers, although only (-)-B-pinene and
(+)-3-carene are found in trees (Mirov, 1961). Myrcene and
terpinolene are achiral.
Synergism between
ethanol and various monoterpenes (or turpentine) is also of
widespread occurrence (Nijholt and Schönherr, 1976; Kohnle, 1985;
Vité et al., 1986; Phillips et al., 1988; Volz, 1988; Schroeder,
1988; Chénier and Philogčne, 1989; Schroeder and Lindelöw, 1989;
Phillips, 1990). These compounds are not only important for primary
attraction to plants, but also may play a role in enhancing the
bark beetles' response to aggregation pheromone (Bedard et al.,
1969, 1970; Pitman et al., 1975; McLean and Borden, 1977; Borden et
al., 1980, 1981; Paiva and Kiesel, 1985; Byers et al., 1988; Miller
and Borden, 1990). Host-tree compounds, ethanol and monoterpenes,
elicited increased entering rates of bark beetles T. lineatum and
P. chalcographus, respectively, into pipe traps baited with
aggregation pheromone (Vité and Bakke, 1979; Bakke, 1983; Byers et
al., 1988). B-Phellandrene (Fig. 4) is slightly attractive alone to
I. pini and enhances response to pheromone (Miller and Borden,
1990), and so the monoterpene might induce entering of holes.
In most of the above studies, the discovery of host compounds
attractive to bark beetles has been by the comparative approach
(similar species are known to be attracted) or by surmising that
identified chemicals in the host would be attractive. Thus, most
studies are incomplete because of the possibility that there are
still undiscovered chemicals important for attraction to the host.
Byers et al. (1985) used the subtractive-combination bioassay and
fractionation method (Byers, 1992b) to rigorously identify the host
volatiles responsible for aggregation of T. piniperda. A
combination of (-)-(S)-alpha-pinene, (+)-(R)-alpha-pinene, (+)-3-carene,
and terpinolene, or each alone, was effective in attracting both
sexes (Fig. 4). During the isolation study, designed for detection
of synergistic pheromone components, no evidence was found for
beetle-produced compounds being attractive, in contrast to most
bark beetles that aggregate en masse on hosts (Byers, 1989a). Byers
et al. (1985) quantified the release rates of alpha-pinene, terpinolene
and 3-carene from a freshly cut log of Scots pine (28 cm x 13 cm
diam.) and found them each to be about 15 mg/day. Release of
comparable amounts in the field competed favorably with a host log
in attracting T. piniperda. They theorized that the attraction to
monoterpenes functioned in the beetle's selection of both host-
species (other common tree species have less monoterpenes) and
recognition of the host's susceptibility (storm-damaged trees are
unable to resist attack and have resinous wounds that release
monoterpenes).
In the isolation of host volatiles attractive to T. piniperda,
a gas-chromatographic adsorbent (Porapak Q), widely used for
trapping insect pheromones, was used to collect headspace air from
the infested pine logs. Unfortunately, Porapak Q will not retain
ethanol molecules due to their small size. Thus ethanol could be a
constituent of the attractive host odor. Vité et al. (1986)
presented evidence that ethanol enhanced the attraction of T.
piniperda to alpha-pinene and terpinolene (identified above) by about
eight-fold, but these results are difficult to confirm since the
chemical release rates were not given. They proposed that ethanol
would be released from diseased trees and thus indicate their
suitability to T. piniperda. Ethanol is attractive when released on
healthy trees since T. piniperda were caught in ethanol-baited
traps on trees, and these beetles also attacked trees baited with
ethanol (Schroeder and Eidmann, 1987; Byers, 1992a). However, the
attraction to ethanol in traps away from trees is weak or
negligible, while monoterpenes in these traps are attractive
(Schroeder, 1988; Schroeder and Lindelöw, 1989; Byers, 1992a).
Ethanol, in fact, sometimes reduces response to attractive
baits. Klimetzek et al. (1986) tested different release rates of
ethanol (24 to 125 mg/day) with an unreported release rate of alpha-
pinene and terpinolene and found that the higher releases of
ethanol inhibited attraction of T. piniperda. However, a control
with ethanol alone, or terpenes alone, was not reported. Schroeder
(1988) increased the release of ethanol in five dosages over an
even wider range from 0 to 50 g/day in combination with a 240
mg/day release of alpha-pinene. In this case, the attraction of T.
piniperda declined linearly with the logarithm of ethanol release,
in conflict with the theory of Vité et al. (1986) that ethanol was
synergistic with monoterpenes.
Schroeder and Lindelöw (1989) provided the first evidence that
could integrate the disparate results. They found that a high
release of alpha-pinene was most attractive to beetles and that ethanol
releases alone from 0 to 3 g/day were barely attractive. At a low
release rate of alpha-pinene (2.4 or 22 mg/day) and thus low
attraction, the lower releases of ethanol from 0 to 3 g/day had a
synergistic effect with alpha-pinene in attracting beetles (Schroeder
and Lindelöw, 1989). Their results are supported by Byers (1992a);
i.e., a weak enhancement of attraction by ethanol at low releases
of the blend of three host monoterpenes, but no observable effect
of ethanol on the greater attraction to higher releases of
monoterpenes.
Ethanol released at even higher rates, 120 mg/day (Klimetzek
et al., 1986) or 50 g/day (Schroeder, 1988), inhibits the response
of T. piniperda to monoterpenes. Therefore, the beetle could find
diseased, but physically uninjured, trees by a weak response to a
synergism between low monoterpene release rates and moderate
ethanol rates - the hypothesis of Vité et al. (1986). These trees
would be tested occasionally by beetles, and if low in resistance
this would permit beetles to continue feeding. Resinosis and
monoterpene release would elicit increased numbers joining in a
mass-
attack. Injured trees with wound oleoresin, and trees under attack
with "pitch tubes", would have a higher monoterpene release and
attract the greatest numbers of beetles, according to Byers et al.
(1985). Trees with high ethanol release rates would indicate a tree
in advanced decay and unsuitable for reproduction, and thus to be
avoided, as theorized by Klimetzek et al. (1986). High monoterpene
releases from trees (freshly wounded and not dead) would not
naturally coincide with high ethanol release rates (presumably
during decay after death). In addition, other compounds such as
verbenone from decaying hosts would inhibit response to
monoterpenes from unsuitable hosts (discussed in the next part).
These studies emphasize the need for releasing semiochemicals at
precise rates during tests in the field. In addition, measurements
of the natural release rates of ethanol and monoterpenes from
various host and nonhost substrates are necessary for further
understanding of bark beetle chemical ecology.

Byers, J.A. 1995. Host tree chemistry affecting colonization in bark
beetles, in R.T. Cardé and W.J. Bell (eds.). Chemical Ecology of
Insects 2. Chapman and Hall, New York, pp. 154-213.
